A self-diagnostic test for Gonorrhea infection. Swab sample required.
Our gonorrhea test kit is a quick and easy way to test for this common STD from the comfort of your own home. The kit comes with everything you need, including a swab for collecting a sample and a dropper for transferring it to the testing strip. Simply follow the instructions provided and wait for your results, which are available in just minutes. This test is highly accurate and provides reliable results, giving you the information you need to seek treatment and protect your sexual health. Whether you are looking to take control of your sexual health or have been experiencing symptoms, our gonorrhea test kit is an effective option.
The Parsagen Home Gonorrhea Test Kit offers you a discreet method of Gonorrhea testing at your own convenience. This is the exact test kit that is being used in government clinics and test labs. Each pack contains a cassette, test tube, micro pipette, 2 swabs, 2 buffer solutions and silicon gel (for keeping contents dry).
Product summary
- Detects Gonorrhea (Neisseria gonorrhoeae) antigen in urogenital samples.
- Not suitable for oral or rectal samples.
- High clinical accuracy. Sensitivity 96.3% specificity: 97.5%.
- For field & health care professional use.
- Tested by certified laboratories worldwide.
- Over 99% accuracy
- TÜV ISO 13485 (ISO’s International Medical Devices Standard) Certified
- GMP (Good Manufacturing Practices) Certified
- Easy to Use
- Total Privacy
Product Description
CATALOG
| Product Name | Specimen | Catalog No. | Quantity per box |
| Gonorrhea Cassette | Swab/Urine | PS-STD-027 | 20T |
KEY POINTS
Detection for: It is as an aid in the early detection of gonococcus infection
Specimen volume: Please refer to the test procedure
Reading time: 10-20 minutes
Relative Sensitivity: 98%;
Relative Specificity: 97.5%.
INTRODUCTION
Gonorrhoea is a sexually transmitted disease. The presence of the bacteria Neisseria gonorrhoeae in body fluids indicates a possible infection.
INTENDED USE
Parsagen® Gonorrhea Cassette Rapid Test Cassette (Swab/Urine) is a rapid immunochromatographic test for the visual detection of Neisseria gonorrhoeae antigen in the secretory specimen from urogenital system and in urine. It is as an aid in the early detection of gonococcus infection.
TEST PROCEDURE
SPECIMEN COLLECTION AND HANDLING
1. Swab Sample:
(1) Use a swab to collect specimen in the following suggested method:
a. Male patients: Swab discharge from the opening of the urinary tract. If no discharge is present, insert the swab 2-3 cm into the urinary tract, gently move 3-5 rings and then take out the cotton swab and put it into sample container.
b. Female patients: Swab discharge from the vaginal opening, then Use cotton swab to insert vagina for 2-4cm and revolve it for 15-30 seconds, then take out the cotton swab and put it into sample container.
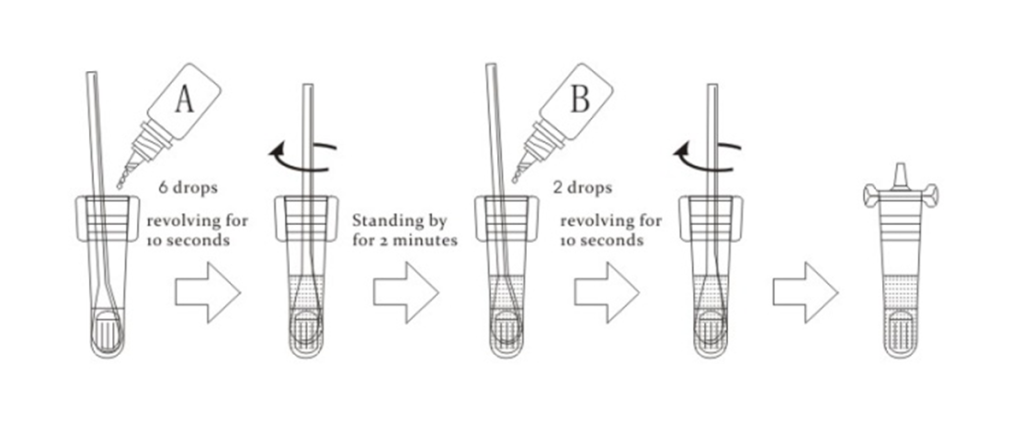
(2) Sample pretreatment
a) Place the swab into the tube and add 6 drops (300μL) specimen extraction Buffer A on the swab, rotate swab and squeeze swab at least 10 seconds.
b) After immobilized for 2 minutes, Add 2 drops (100μL) extraction Buffer B into the tube and mix well. Revolve the swab quickly for at least 10 seconds , finally squeeze liquid in the swab head as more as possible. Discard the swab into a disinfectant container. .
c) Specimen collected in the dilution should be stored at 4-8°C and tested within 24 hours. If specimens are not going to be tested with 24 hours, they should be stored at -20°C or below.
2. Urine Specimen:
Please note: one individual found this test not to be senstive enough for urine samples.
We advise you to use swab specimens if at all possible. A urine specimen must be collected in a clean and dry container. Transfer the specimen to a centrifuge tube and centrifuge. Pour off the urine and treat the urine sediment as the sample.
ASSAY PROCECURE
1. Preparation
(1)Allow the test device, the specimen and/or controls to equilibrate to room temperature (15-30℃) prior to testing.
(2)When you are ready to begin testing, open the sealed pouch by tearing along the notch.
2. Testing
(1)Remove the test cassette from the pouch and use it as soon as possible.
(2)Place the testing device on a flat surface.
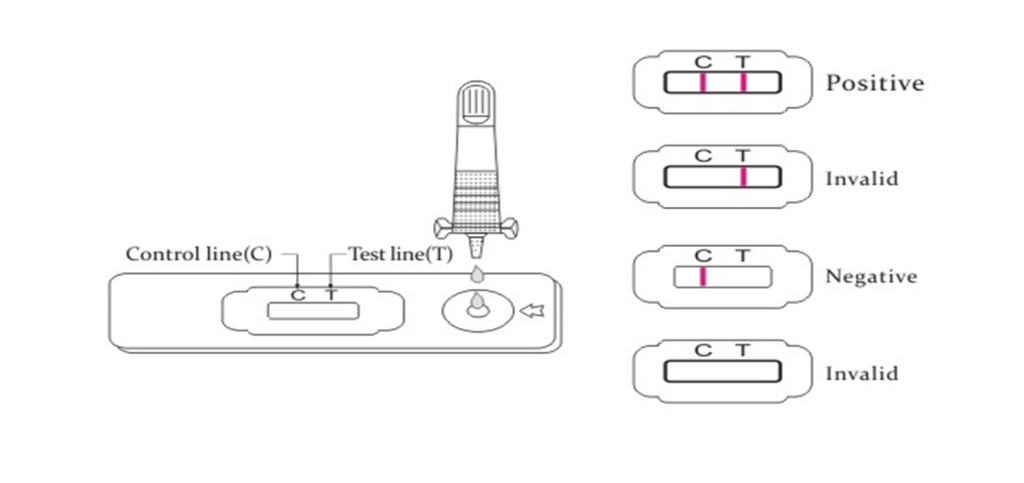
(3)Holding the sample dropper vertically, carefully dispense exactly 2 drops of the specimen into the specimen port (S) of the cassette,and then start the timer. Avoid trapping air bubbles in the specimen well.
Alternatively, using a micropipette, carefully dispense exactly 80~100μl of the specimen into the specimen port (S).
(4)Read the result between 10-20 minutes. Do not read results after 30 minutes.
PERFORMANCE CHARACTERISTICS
Expected Values
The Test is designed to show a positive result for Neisseria gonorrhoeae at a concentration of 1 x 105 bacteria/ml.
Negative results are expected in healthy non-infected women and men.
Accuracy
130 patients were tested for gonorrheal infection and their swab specimens were tested in parallel using Parsagen® Gonorrhea Cassette Rapid Test Cassette (Swab/Urine) and a conventional culture test. For calculating sensitivity and specificity, the cell culture result is assumed to be 100% accurate. The results are shown as follow:
| Cell Culture | Gonorrhea Test cassette | |
| Test Positive | Test Negative | |
| True Positive | 48 | 2 |
| True Negative | 1 | 79 |
Relative Sensitivity: 98%;
Relative Specificity: 97.5%.
3. Interfering Substances
The cross reactivity of product was evaluated using other bacterium strains. E. Coli, Salmonella Typhi, Staphylococcus Aureus, Pseudomonas Aeruginosa, Shigella, and Proteus were added to samples and were tested for cross reactivity. No cross reactivity was observed.
LIMITATIONS
1. The test is used for testing secretion from male urethra and female cervices. Reliability of other samples have not been certified.
2. The test is only used for qualitative detection of gonococcus antigen existence, concentration of gonococcus antigen cannot be confirmed.
3. The test result should be used in conjunction with other clinical information such as clinical signs and symptoms and other tests to diagnose gonorrhoea. A positive result would need a further confirmatory test.
4. The Test is a presumptive, screening test for the presence of Neisseria gonorrhoeae. If test results are negative but clinical symptoms are indicative of gonorrhoeal infection, further tests are recommended. Cell culture is the standard reference test method for the detection of Neisseria gonorrhoeae.
5. As with all diagnostic tests, a definitive clinical diagnosis should not be based on the result of a single laboratory test, but should only be made by the physician after all clinical and laboratory tests have been evaluated.
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.
