Our herpes 2 blood home test kit is a quick and easy way to test for HSV-2 (genital herpes) in the comfort of your own home. The kit comes with everything you need, including a finger-prick blood test and a dropper for transferring a sample to the testing strip. Simply follow the instructions provided and wait for your results, which are available in just minutes. This test is highly accurate and provides reliable results, giving you the information you need to seek treatment and protect your sexual health. Whether you are looking to take control of your sexual health or have been experiencing symptoms, our herpes 2 blood home test kit is an effective option.
The Parsagen Herpes-2 Test Kit offers you a discreet method of Herpes-2 testing at your own convenience. This is the exact test kit that is being used in government clinics and test labs. Each pack contains a cassette, micro pipette, alcohol preparation pad, diluent bottle (chemical), lancet (needle) and silicon gel (for keeping contents dry).
Product summary
- Detects Herpes Simplex 2 (Anti-HSV 2 IgG/IgM).
- For field & health care professional use.
- Tested by certified laboratories worldwide.
- High clinical accuracy. Sensitivity: 98.04% specificity: 98.3%.
- –TÜV ISO 13485 (ISO’s International Medical Devices Standard) Certified
- GMP (Good Manufacturing Practices) Certified
- Easy to Use
- Total Privacy
Product Description
CATALOG
| Product Name | Specimen | Catalog No. | Quantity per box |
| Herpes Simplex 2 Cassette | Serum/Plasma | PS-STD-025 | 15T |
KEY POINTS
Detection for: IgM antibodies to Herpes simplex virus-2 (HSV-2) in serum or plasma;
Reading time: 15 minutes;
Sensitivity & Specificity & Accuracy:
Sensitivity: 90.0% (95%CI*: 68.3%~98.8%)
Specificity: 100% (95%CI*: 98.5%~100.0%)
Accuracy: 99.1 %( 95%CI*: 96.8%~99.9%)
*Confidence Interval
INTRODUCTION
Herpes Simplex Virus 2 is a infectious disease that, while infecting the pregnant women, may cause birth defects in their newborns. The infections usually cause few, if any, symptoms in the pregnant woman, but pose greater risks of serious birth defects for neonates.
INTENDED USE
Parsagen® HSV-2 Cassette (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of IgM antibody to Herpes simplex virus-2 (HSV-2) in serum or plasma.
TEST PROCEDURE
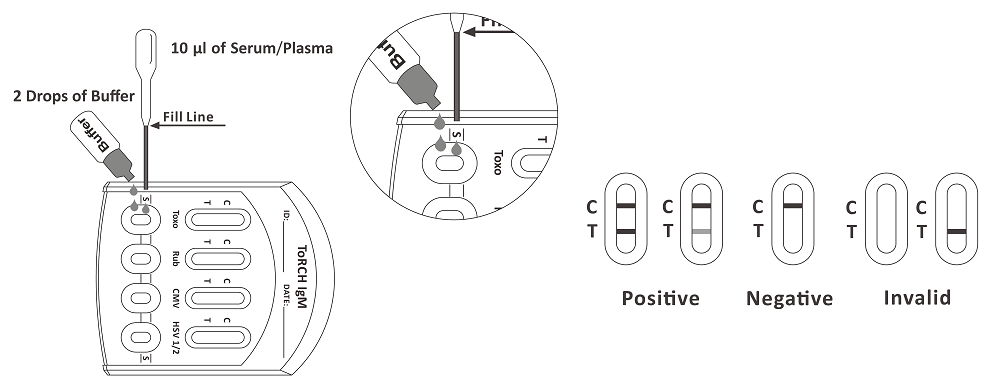
1. Bring the pouch to room temperature before opening it. Remove the test cassette from the sealed pouch and use it as soon as possible. Best results will be obtained if the assay is performed within one hour.
2. Place the test cassette on a clean and level surface. Hold the dropper vertically and draw the serum/plasma up to the Fill Line as shown in illustration below (approximately 10 μl ) to the S area of each specimen well of the test cassette, then add 2 drops of buffer (approx. 80 μl) respectively and start the timer. Avoid trapping air bubbles in the specimen well. See the illustration below.
3. Wait for the colored line(s) to appear. The result should be read at 15 minutes. Do not interpret results after 20 minutes
PERFORMANCE CHARACTERISTICS
Sensitivity and Specificity
Parsagen® HSV-2 Cassette (Serum/Plasma) was compared with leading commercial tests; the results show that Parsagen® HSV-2 Cassette (Serum/Plasma) has a high sensitivity and specificity for each of its sections.
| Method | HSV 1/2 EIA (IgM) | Total Results | ||
| HSV-2 IgM Test Cassette(Serum/Plasma) | Results | Positive | Negative | |
| Positive | 18 | 0 | 18 | |
| Negative | 2 | 200 | 202 | |
| Total Results | 20 | 200 | 220 | |
*Confidence Interval
Precision
Intra-Assay
Within-run precision has been determined by using 15 replicates of the specimens containing negative, low positive, middle and high positive of HSV-2. The negative, low positive, and high positive values were correctly identified >99% of the time.
Inter-Assay
Between-run precision has been determined by using the specimens of negative, low positive, middle and high positive of HSV-2 in 15 independent assays. Three different lots of the HSV-2 IgM Rapid Test Cassette (Serum/Plasma) have been tested using negative, low positive, middle and high positive specimens. The specimens were correctly identified >99% of the time.
Cross-reactivity
Parsagen® HSV-2 Cassette (Serum/Plasma)has been tested for HAV+, HBV+, HIV+, HCV+, HEV+, Syphilis+, HAMA+, RF+, H.pylori+, MONO+, CEA and AFP. The results showed no cross-reactivity. In addition, the rubella, CMV and HSV-2 IgM positive for Toxo section, the Toxo, CMV and HSV-2 IgM positive for Rubella section, the Toxo, Rubella and HSV-2 IgM positive for CMV section and the Toxo, Rubella and CMV IgM positive for HSV-2 section showed no cross-reactivity.
Interfering Substances
The following compounds have also been tested using Parsagen® HSV-2 Cassette (Serum/Plasma) and no interference was observed.
Ascorbic Acid: 20mg/ml
Hemoglobin: 1000mg/dl
Gentisic Acid: 20 mg/dL
Oxalic Acid: 60mg/dL
Bilirubin: 1000mg/dL
Uric acid: 20mg/ml
Acetaminophen: 20 mg/dL
Aspirin: 20mg/dl
Methanol: 10%
Creatine: 200mg/dl
Albumin: 2 g/dL
Caffeine: 20 mg/dl
LIMITATIONS
1. Parsagen® HSV-2 Cassette (Serum/Plasma) is for in vitro diagnostic use only. This test should be used for detection of IgM antibodies to HSV 1/2 in serum or plasma specimens. Neither the quantitative value nor the rate of increase in the concentration of IgM antibodies to HSV-2 can be determined by this qualitative test.
2. Parsagen® HSV-2 Cassette (Serum/Plasma) will only indicate the presence of IgM antibodies to HSV-2 in the specimen and should not be used as the sole criteria for the diagnosis of HSV-2 infections for which the positive result is obtained.
3. As with all diagnostic tests, all results must be considered with other clinical information available to the physician.
4. If the test result is negative and clinical symptoms persist, additional follow-up testing using other clinical methods is suggested. A negative result for any one out of the four infections of HSV-2 at any time does not preclude the possibility of that particular infection
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.
